PRIN 2022 PNRR / Silvia Todros

Acronimo: 3D-L-INKED
Titolo: Novel visible light-degradable hydrogels embedded with cell-laden proteinosomes for the 3D bioprinting of diaphragm-like muscle tissues
Responsabile scientifico: dott.ssa Silvia TODROS - Dipartimento di Ingegneria Industriale-Università' degli Studi di PADOVA
Coordinatore: prof. Pierangelo GOBBO - Università degli Studi di TRIESTE
Partner-Unità di ricerca: Dipartimento di Ingegneria Industriale-Università degli Studi di PADOVA
Bando: PRIN 2022 - Decreto Direttoriale n. 1389 del 01/09/2023
Durata: 30/11/2023 - 29/11/2025 (24 mesi)
Budget totale progetto: € 235.500,00
Abstract del progetto
Congenital diaphragmatic hernia (CDH) is a rare birth defect that implies an incomplete development of the diaphragmatic muscle involving the herniation of abdominal organs in the chest. This research project aims to introduce a disruptive new strategy for the fabrication of 3D printed, myoblast-laden, patient-specific patches for the potential treatment of postnatal CDH (Figure 1).
Although attempts have been reported to 3D bioprint constructs that mimic muscular tissue, their clinical translation has not been achieved yet, since these constructs consist of cells dispersed in a non-clinically approved hydrogel matrix which cannot be ultimately implanted in the body. In order to solve this key issue, 3D-L-INKED will introduce a novel chemically crosslinked hydrogel acting as the support for bioprinting and tissue growth that can be gradually degraded using visible light and, ultimately, removed completely upon tissue maturation. Using this new visible light strategy, we will be able to effectively remove the unwanted hydrogel component before tissue implantation without damaging the cells.
Another important challenge of bioprinting is to reduce the high shear and compressive stress that is exerted on the cells during their extrusion through the printer nozzles. To address this challenge, we will pioneer the encapsulation of cells inside soft, semipermeable and biocompatible microcompartments called "proteinosomes" that will function as protective "shells'' against mechanical stress resulting from bioprinting. Importantly, in the initial phase of the tissue culture, the nutrients and gasses will be able to permeate from the external environment through the proteinosome membrane, whereas in the later stage the soft proteinosome membrane will be gradually torn apart by the growing tissue.
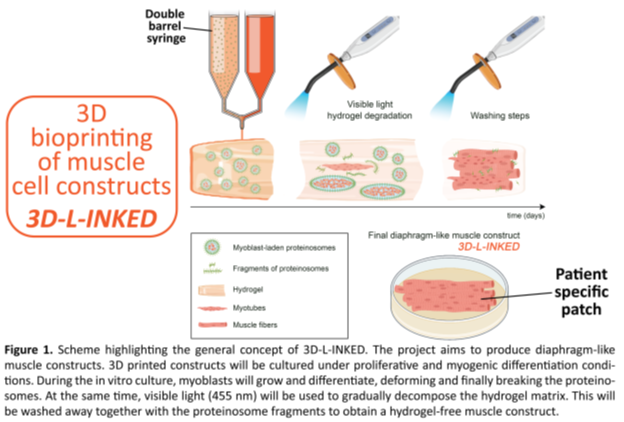
Overall, 3D-L-INKED will allow for the fabrication of precise, patient-specific tissue constructs that will have a broad impact not only in the treatment of CDH, but also on a variety of other muscular congenital defects. In fact, seen the high modularity of our approach (possibility of using different cell lines, fine-tuning of hydrogel properties and of shape and size of the tissue-like constructs), we will be able to apply the strategy to multiple tissue engineering approaches and regenerative medicine challenges.



